Acetal protecting groups protect carbonyls from bases, nucleophiles and hydride reduction. Among many variants, most common are dimethyl acetals, 1,3-dioxolanes and 1,3-dioxanes.
There is no single acetal protecting group! Rather, this is a broader family of similar protecting groups.
➡️ This article explains properties and mechanisms of acetals (protection and deprotection).
What Are Acetal Protecting Groups?
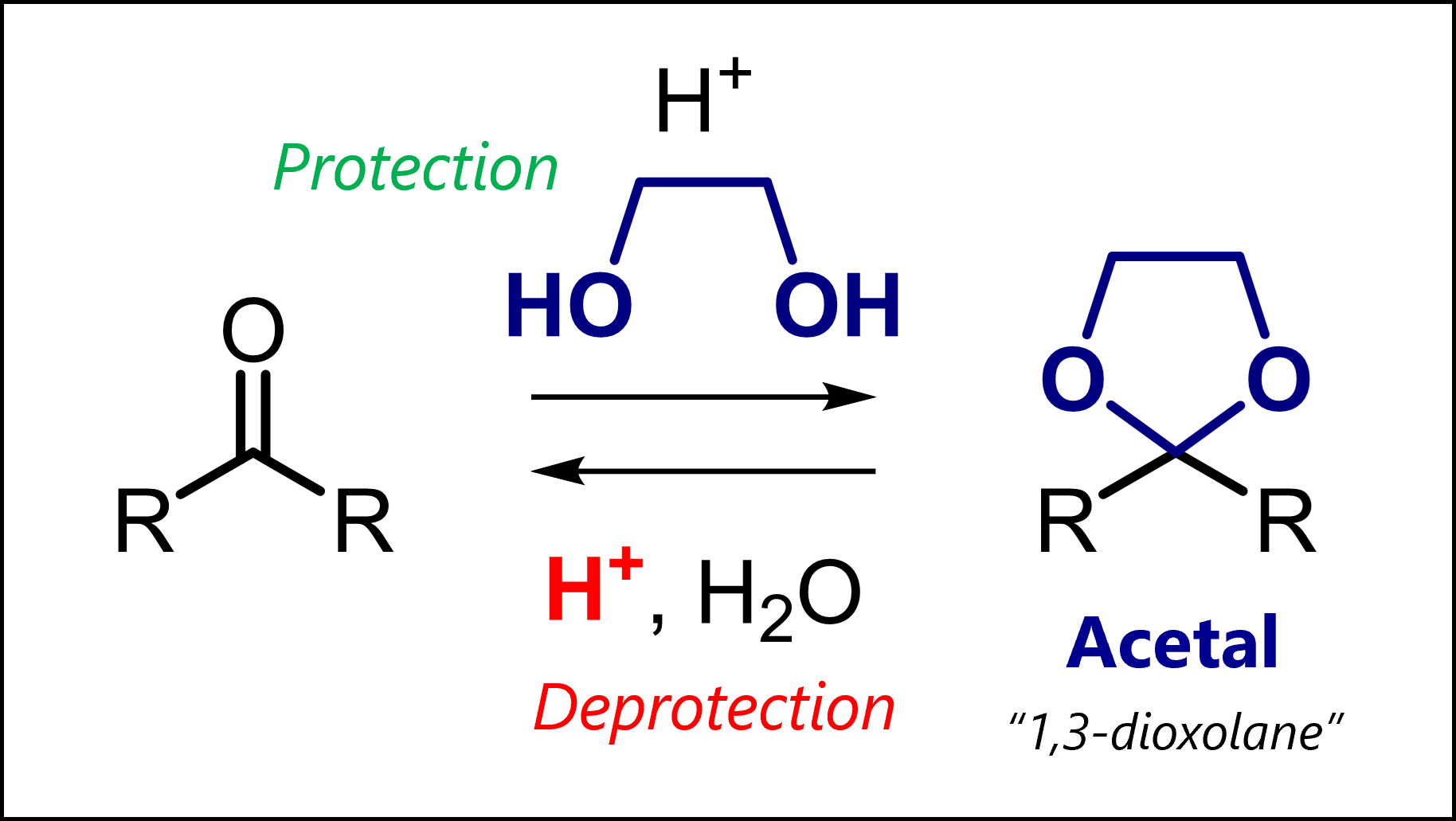
Acetals always protect carbonyl compounds. But how? This is where the variety can come from. On one hand, acyclic acetals form by reaction with an alcohol (-OH) or thiol (-SH) and catalytic acid. On the other, cyclic acetals form when carbonyls react with a diol or dithiol and catalytic acid.
Do you already know if cyclic or acyclic acetals are more stable? Why? (see below)
You will know that carbonyls are nucleophilic at the carbon. Any acetal protecting group renders it stable to these nucleophiles: aqueous and non-aqueous bases, organometallic reagents and hydrides. As always, we want to avoid unwanted reactions of one group (here, the carbonyl) to instead perform chemistry at another functional group. As we will see below, formation of acetals involves a two-step mechanism, including nucleophilic attack and subsequent dehydration, which drives the equilibrium towards product formation.
Difference between Acetal and Ketal
You might not be aware, but back in the day, people used to separate acetals – made from aldehydes – and ketals – made from ketones. Nowadays, acetal is the umbrella term that describes both – while ketal remains restricted to ketones (link to IUPAC definition).

Types Of Acetal Protecting groups
As mentioned, there are several relatives in the acetal protecting group family. The good thing is that they work very similarly!
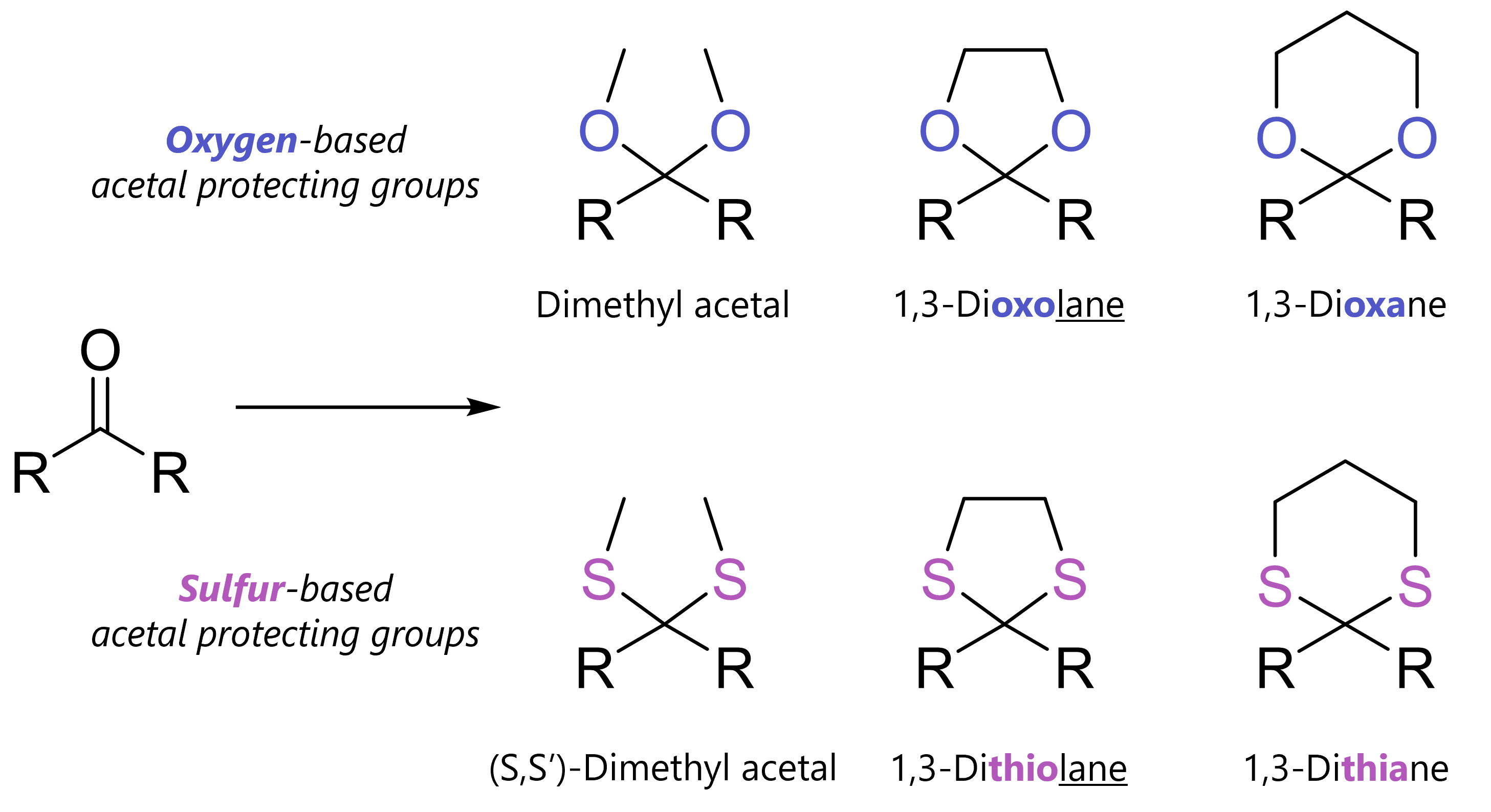
=> You should simply know that acetals can be oxygen-based or less commonly, sulfur-based. The simplest acyclic acetal is the dimethyl acetal. Cyclic acetals have five-membered rings (1,3-dixolane; 1,3-dithiolane) six-membered rings (1,3-dioxane, 1,3-dithiane).
Acetal protection mechanism
As an example, 1,3-dioxolanes are prepared by treating carbonyls with ethylene glycol and acid.
=> Acetal protection or acetalization requires catalytic acid to activate the carbonyl (but only catalytic because the proton is regenerated in the final step)
=> Acetalization is a condensation as the original oxygen is kicked out as water

Typical conditions: Ethylene glycol and cat. TsOH (acid) in C6H6 as solvent at reflux.
Because every reaction is an equilibrium (imagine the arrows also going from right to left), chemists use ways to remove water from the reaction to ensure it can’t react back. For acetalizations, this involves using a Dean-Stark trap. The Dean-Stark trap is a glassware that collects water formed in a reaction through an azeotropic distillation. You might have heard about it – if not, does not matter.
This is a physical removal – alternatively, dehydrating agents like trimethyl orthoformate can chemically remove the water by reacting with it (“scavenger”).
The same mechanism applies if we use other diols (e.g., to form six-membered 1,3-dioxanes), alcohols (e.g., methanol to form dimethyl acetals) or thiols to form sulfur-based acetal protecting groups.
Acetal Deprotection Mechanism
Deprotecting acetals is very similar to introducing them!
The most common is an acid-catalyzed hydrolysis. Again, make sure you understand why it only requires catalytic acid (i.e., less than 1 “equivalent” of moles).

Typical conditions: Cat. pyridinium tosylate PPTS or HCl (as the acid) in a mixture of water (for the deprotection) and an organic solvent (to dissolve the starting material).
The sulfur-based acetals are special as they can also be removed with heavy metal salts – so Lewis acids like mercury(II) or silver(I) – or oxidants. The oxygen-based acetals are stable to these conditions.
We will go into 1,3-dithianes and 1,3-dithiolanes into more detail in another post.

Tired of serious chemistry?
Take a break with “Periodic Tales – The Freshman Mole”, a satirical novel that’s the opposite of educational.
Dedicated to every chemistry and STEM student who asked: “Why did no one warn me?”
Acetal protecting group stability
Are cyclic or acyclic acetals more stable?

Cyclic acetals are more stable than acyclic ones. Why? Acidic hydrolysis starts with protonation (catalytic acid), and goes via the oxonium intermediate.
For the cyclic acetal, the newly released hydroxyl group is still in the same molecule – so the reverse reaction would be an intramolecular reaction which is very fast (entropically favored).
For acyclic acetals, formation of the oxonium cleaves off an alcohol as a separate molecule. Because the deprotection is in aqueous solvent, we have a lot of water molecules around. It is now much more likely water will attack the oxonium (leading to deprotection of the carbonyl) instead of the alcohol attacking. This is because we only have 1 molecule of alcohol formed for 1 molecule of starting material; on the other hand, we have a large excess of water molecules.
Learn more about other protecting groups, or check out my educational videos for interdisciplinary and advanced content!
Acetal Protecting Group References
- P. G. M. Wuts, T. W. Greene: Greene’s Protective in Organic Synthesis (Wiley)

Leave a Reply
You must be logged in to post a comment.